Introduction
Sampling compliance and net content control are critical components of quality systems in regulated industries such as food and beverage manufacturing. The U.S. Food and Drug Administration (FDA) requires manufacturers to ensure that labeled net contents are accurate and not misleading (21 CFR Part 101). Ongoing in-process checks and documented sampling are essential to demonstrate control of fill weights, volumes, and other net content attributes.
By enforcing time-based sampling frequencies, tracking startup and production activities, and generating compliance reports, DataLyzer Qualis helps manufacturers demonstrate adherence to FDA net content requirements and internal quality standards. The system provides objective evidence that measurements were taken at the right time, in the right quantity, and under the correct production conditions.
Configuration of Compliance Management
When a production line is not running, sampling is normally not required. Samples are only required when all of the following conditions are met:
- There is an active shift
- There is an active product
- The line is running or in startup mode
To support this, DataLyzer Qualis SPC allows the system to be configured with machines, products, activities, and shifts. This configuration ensures that sampling requirements are only enforced when production conditions require them, reducing unnecessary data collection while maintaining compliance.
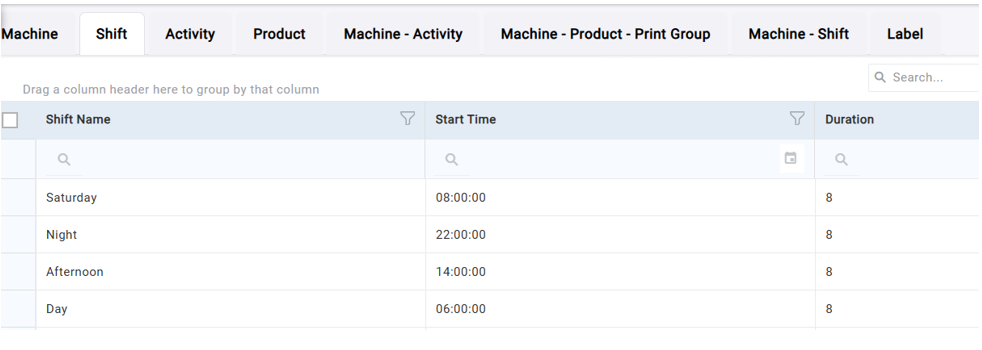
Figure 1: Configuration of machines, products, activities and shifts.
On each manufacturing line, many potential measurements can be taken. Because control charts may differ by product, DataLyzer Qualis SPC allows you to define which control charts apply to each product running on a specific line. These charts are then included in the sampling compliance report only when the relevant product is active.
Each control chart can also have a defined sampling frequency during normal production. During startups, however, sampling requirements may differ. For example, only a single subgroup may be required during startup, or the sampling frequency may be higher than during steady-state production. In DataLyzer Qualis, two different startup modes can be configured, such as daily startup and shift change.
During production, operators can indicate whether the line is:
- Running production
- In one of the defined startup modes
- Down (no measurements required)
For each quality characteristic, sampling frequencies can be configured based on the machine’s status. In the operator information screen, the sampling compliance requirements for each characteristic are clearly defined.

Figure 2: Setup of Sampling Frequency
Sampling intervals are defined in minutes (for example, every 30 minutes during production). For startup phases such as daily startup or shift change, a time-based interval may not always be known. In these cases, the requirement is often stated that at least one measurement must be taken before switching to normal production.
A mandatory checkbox indicates whether a measurement is required for a specific characteristic during a given activity. For example, at least one measurement may be required during daily startup, while no measurement may be required during a shift change.
During production, operators select the active product and the current machine activity. Based on this information and the configured sampling frequencies, Qualis SPC automatically indicates which measurements need to be taken and alerts the operator when a measurement is due or nearly overdue. This provides clear, real-time guidance to operators regarding inspection plans.
Sampling Compliance Report
A Sampling Compliance Report can be generated for one or more machines.
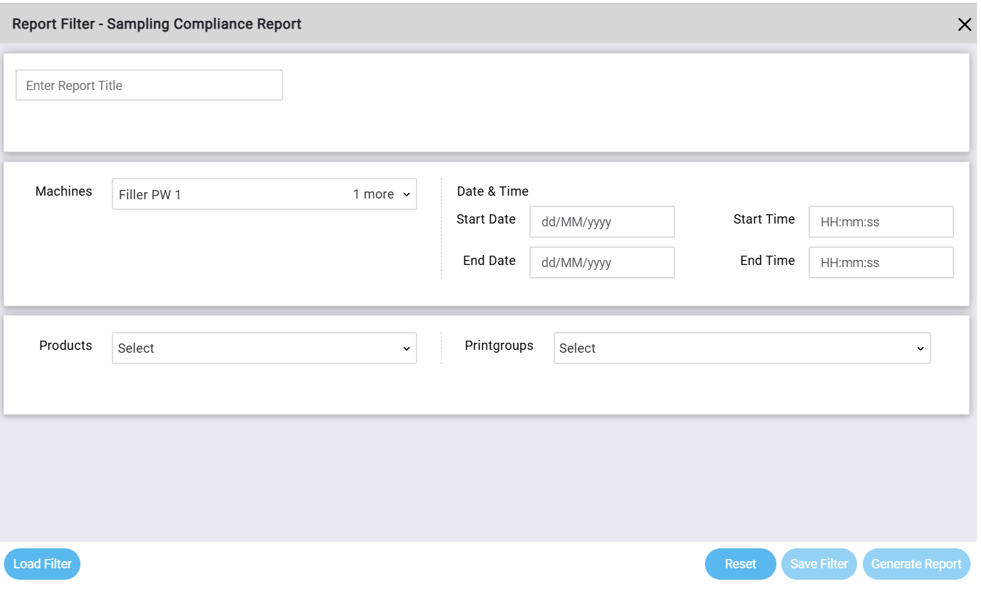
Figure 3: Sampling Compliance Report Filter
To run the report, the user selects:
- The machines to include
- The date and time range for the report
Once the machines and time period are selected, Qualis SPC identifies all products that were active on those machines during the selected timeframe. This results in a complete list of products that were produced. The user can include all products or select specific products in the report.
Next, the printgroup selection displays the relevant control charts (quality characteristics) associated with the selected machines and products. These charts define which measurements are evaluated in the report.
After selecting Generate Report, the sampling compliance report is produced.
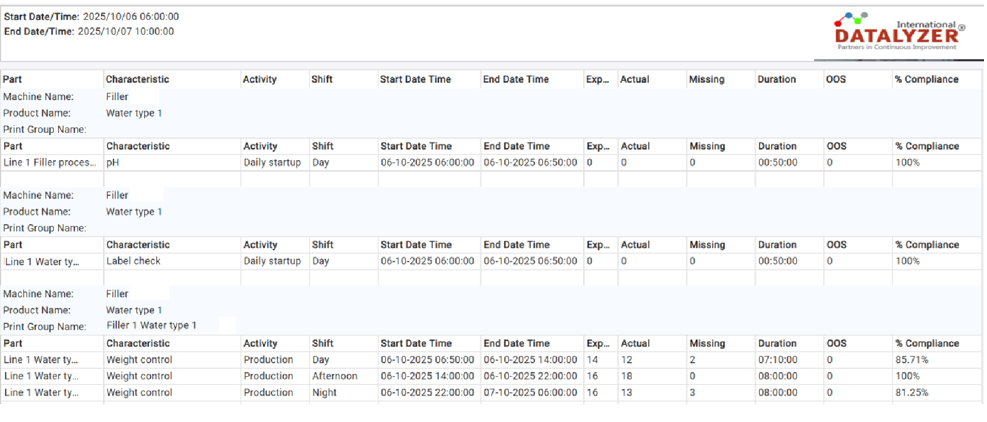
Figure 4: Example Sampling Compliance Report
The report shows, for each machine activity:
- The quality characteristics that should have been measured
- The expected number of subgroups
- The actual number of subgroups collected
- The number of missing subgroups
- The number of out-of-spec measurements
- The overall percentage of sampling compliance
Missing measurements are evaluated by count and by timing. For example, if a subgroup is required every 30 minutes during a five-hour production run, 10 subgroups are expected. If all measurements are taken within the first 10 minutes, the report will still indicate 9 missing subgroups, because the required sampling intervals were not met.
This approach ensures that compliance reflects real-world production monitoring rather than just total measurement counts.
Summary
DataLyzer Qualis, the leading web-based SPC solution for the food and beverage industries, now offers a comprehensive sampling compliance reporting capability based on actual production activities.
The system evaluates sampling compliance across machines, shifts, activities, and defined sampling frequencies. It guides operators through their inspection requirements and provides supervisors and quality managers with clear reporting to verify that sampling was performed according to defined requirements. This supports both operational discipline and regulatory compliance, including FDA net content control requirements.
Discover how DataLyzer and our Qualis 4.0 SPC software can help you to implement, amongst others, Sampling Compliance Reporting. Our team of experts is ready to show you how our webbased solution can be tailored to your needs.



